NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-.

LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet].
Show detailsOVERVIEW
Introduction
Clemastine is a first generation antihistamine that is used for symptoms of allergic rhinitis and the common cold. Clemastine has not been linked to instances of clinically apparent acute liver injury.
Background
Clemastine (kle mas' teen) is a first generation antihistamine that is used for alleviation of symptoms of allergic rhinitis, the common cold and allergic urticaria, including sneezing, cough, runny note, watery eyes and itching. Clemastine belongs to the ethanolamine class of antihistamines (with diphenhydramine and dimenhydrinate) and was first approved for use in the United States in 1977. Clemastine is available in multiple generic forms as tablets and syrup, many of which are available without prescription. Common brand names include Tavist Allergy and Dayhist Allergy. The recommended adult oral dose ranges from 1 to 2 mg one to three times daily. Common side effects include sedation, impairment of motor function, confusion, dizziness, blurred vision, dry mouth and throat, palpitations, tachycardia, abdominal distress, constipation and headache. Antihistamines can worsen urinary retention and glaucoma.
Hepatotoxicity
Despite widespread use, the first generation antihistamines such as clemastine have rarely been linked to liver test abnormalities or to clinically apparent liver injury. The reason for their safety may relate to low daily dose and limited duration of use.
Likelihood score: E (unlikely to be a cause of clinically apparent liver injury).
References on the safety and potential hepatotoxicity of antihistamines are given together after the Overview section on Antihistamines.
Drug Class: Antihistamines
PRODUCT INFORMATION
REPRESENTATIVE TRADE NAMES
Clemastine – Generic, Dayhist Allergy®, Tavist Allergy®
DRUG CLASS
Antihistamines
Product labeling at DailyMed, National Library of Medicine, NIH
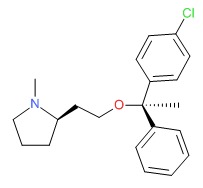
CHEMICAL FORMULA AND STRUCTURE
| DRUG | CAS REGISTRY NUMBER | MOLECULAR FORMULA | STRUCTURE |
|---|---|---|---|
| Clemastine | 15686-51-8 | C21-H26-Cl-N-O |
 |
- Effectiveness of clemastine fumarate for treatment of rhinorrhea and sneezing associated with the common cold.[Clin Infect Dis. 1997]Effectiveness of clemastine fumarate for treatment of rhinorrhea and sneezing associated with the common cold.Turner RB, Sperber SJ, Sorrentino JV, O'Connor RR, Rogers J, Batouli AR, Gwaltney JM Jr. Clin Infect Dis. 1997 Oct; 25(4):824-30.
- Comparative study of the efficacy and tolerance of terfenadine and clemastine in patients with seasonal allergic rhinitis.[Arzneimittelforschung. 1982]Comparative study of the efficacy and tolerance of terfenadine and clemastine in patients with seasonal allergic rhinitis.Gastpar H, Dieterich HA. Arzneimittelforschung. 1982; 32(9a):1211-3.
- A multicentre study of loratadine, clemastine and placebo in patients with perennial allergic rhinitis.[Allergy. 1990]A multicentre study of loratadine, clemastine and placebo in patients with perennial allergic rhinitis.Frølund L, Etholm B, Irander K, Johannessen TA, Odkvist L, Ohlander B, Weeke B. Allergy. 1990 May; 45(4):254-61.
- Review Carbinoxamine.[LiverTox: Clinical and Researc...]Review Carbinoxamine.. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. 2012
- Review Doxylamine.[LiverTox: Clinical and Researc...]Review Doxylamine.. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. 2012
- Clemastine - LiverToxClemastine - LiverTox
Your browsing activity is empty.
Activity recording is turned off.
See more...