Summary
Clinical characteristics.
FRMD7-related infantile nystagmus (FIN) is characterized by either the onset of horizontal, conjugate, gaze-dependent nystagmus in the first six months of life or periodic alternating nystagmus (with cyclical changes of nystagmus direction) of infantile onset. Binocular vision and color vision are normal and visual acuity is typically better than 6/12. An abnormal head posture is seen in approximately 15% of affected individuals. The eyes are structurally normal and electrophysiologic studies (e.g., visual evoked potential, electroretinogram) are normal. Affected females report slightly better visual acuity than affected males; however, no differences between males and females in the amplitude, frequency, and waveform of nystagmus are observed.
Diagnosis/testing.
The diagnosis is based on clinical findings (including, when possible, ocular motility recordings). Identification of a hemizygous (in a male proband) or heterozygous (in a female proband) pathogenic variant in FRMD7 by molecular genetic testing can establish the diagnosis if clinical features are inconclusive.
Management.
Treatment of manifestations: Correction of refractive errors as early as possible. Prisms may be useful in those with binocular vision whose nystagmus is dampened by convergence. Memantine and gabapentin can improve visual acuity, intensity of nystagmus, and foveation. Surgical approaches include: the Anderson-Kestenbaum procedure (surgery of the extraocular muscles to shift the null zone to the primary position) in order to correct anomalous head posture and horizontal rectus tenotomy to improve the waveform of the nystagmus and visual function.
Surveillance: Regular monitoring, especially during childhood, to evaluate visual acuity and development of refractive errors, strabismus, and/or amblyopia.
Genetic counseling.
FIN is inherited in an X-linked manner. Affected males transmit the pathogenic variant to all of their daughters and none of their sons. Women who are carriers have a 50% chance of transmitting the pathogenic variant in each pregnancy. Carrier testing for at-risk female relatives and prenatal testing for a pregnancy at increased risk are possible once the pathogenic variant has been identified in the family.
Diagnosis
Currently there are no formal diagnostic clinical criteria for FRMD7-related infantile nystagmus.
Suggestive Findings
The diagnosis of FRMD7-related infantile nystagmus (FIN) should be suspected in a proband with the following findings [Thomas et al 2008].
Clinical findings
- Onset of nystagmus during infancy (age ≤6 months)
- Horizontal and conjugate nystagmus oscillations
- Visual acuity that is typically better than 0.3 LogMAR (Snellen equivalent 6/12)
- Good binocular vision and normal color vision
- Family history of nystagmus consistent with X-linked inheritance
- In some individuals: dampening of the nystagmus by convergence
- Less common findings: anomalous head posture (15% of affected individuals) and strabismus (8% of affected individuals)
Optical coherence tomography. Normal foveal morphology on optical coherence tomography or (less commonly) grade 1 foveal hypoplasia [Thomas et al 2014]
Eye movement recordings
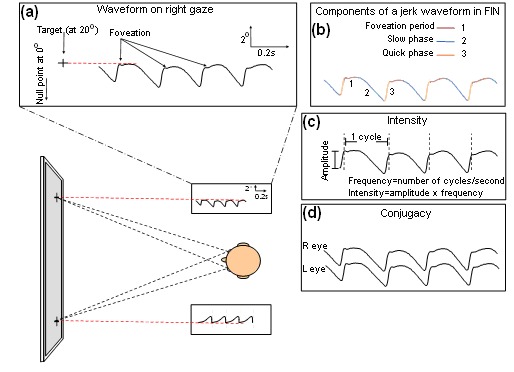
- Affected individuals typically exhibit a pendular or jerk-related waveform with horizontal and conjugate oscillations. In the jerk waveform, the slow phase has an increasing velocity.
- Amplitude of nystagmus can be gaze dependent (i.e., small amplitude on central gaze when compared to left and right gaze).
- Amplitude and direction of the quick phase may be time dependent (periodic alternating nystagmus) [Thomas et al 2011].
Other evaluations. The following are normal in individuals with FIN:
- Slit-lamp biomicroscopy (normal iris pigmentation with no iris transillumination)
- Fundoscopy
- Cranial MRI
- Electroretinogram
- Visual evoked potentials
Establishing the Diagnosis
Male proband. The diagnosis of FRMD7-related infantile nystagmus (FIN) is suspected in a male proband with the above Suggestive Findings. Diagnosis is established by identification of a hemizygous pathogenic (or likely pathogenic) variant in FRMD7 by molecular genetic testing (see Table 1).
Female proband. The diagnosis of FIN is suspected in a female proband with the above Suggestive Findings. Diagnosis is established by identification of a heterozygous pathogenic (or likely pathogenic) variant in FRMD7 by molecular genetic testing (see Table 1).
Note: (1) Per ACMG/AMP variant interpretation guidelines, the terms "pathogenic variants" and "likely pathogenic variants" are synonymous in a clinical setting, meaning that both are considered diagnostic and both can be used for clinical decision making [Richards et al 2015]. Reference to "pathogenic variants" in this section is understood to include any likely pathogenic variants. (2) Identification of a hemizygous or heterozygous FRMD7 variant of uncertain significance does not establish or rule out the diagnosis.
Molecular genetic testing approaches can include a combination of gene-targeted testing (single-gene testing, multigene panel) and comprehensive genomic testing (exome sequencing, genome sequencing) depending on the phenotype.
Gene-targeted testing requires that the clinician determine which gene(s) are likely involved, whereas genomic testing does not. Because the phenotype of FIN is broad, individuals with the distinctive findings described in Suggestive Findings are likely to be diagnosed using gene-targeted testing (see Option 1), whereas those in whom the diagnosis of FIN has not been considered are more likely to be diagnosed using genomic testing (see Option 2).
Option 1
When the phenotypic findings suggest the diagnosis of FIN molecular genetic testing approaches can include single-gene testing, or use of a multigene panel:
- Single-gene testing. Sequence analysis of FRMD7 detects small intragenic deletions/insertions and missense, nonsense, and splice site variants; typically, exon or whole-gene deletions/duplications are not detected.Perform sequence analysis first. If no pathogenic variant is found perform gene-targeted deletion/duplication analysis to detect intragenic deletions or duplications.
- A multigene panel that includes FRMD7 and other genes of interest (see Differential Diagnosis) is most likely to identify the genetic cause of the condition while limiting identification of variants of uncertain significance and pathogenic variants in genes that do not explain the underlying phenotype. Note: (1) The genes included in the panel and the diagnostic sensitivity of the testing used for each gene vary by laboratory and are likely to change over time. (2) Some multigene panels may include genes not associated with the condition discussed in this GeneReview. (3) In some laboratories, panel options may include a custom laboratory-designed panel and/or custom phenotype-focused exome analysis that includes genes specified by the clinician. (4) Methods used in a panel may include sequence analysis, deletion/duplication analysis, and/or other non-sequencing-based tests.
Option 2
When the diagnosis of FIN is not considered because an individual has atypical phenotypic features, comprehensive genomic testing (which does not require the clinician to determine which gene[s] are likely involved) is the best option. Exome sequencing is most commonly used; genome sequencing is also possible.
For an introduction to comprehensive genomic testing click here. More detailed information for clinicians ordering genomic testing can be found here.
Table 1.
Molecular Genetic Testing Used in FRMD7-Related Infantile Nystagmus
Clinical Characteristics
Clinical Description
Affected individuals usually develop nystagmus within the first six months of life; the mean age of onset is two months. Nystagmus can be gaze-dependent oscillations or time-dependent oscillations (periodic alternating nystagmus) [Thomas et al 2008, Thomas et al 2011].
Nystagmus waveform characteristics are established by eye movement recordings that assess the following (see Figure 1 and Figure 2):

Figure 2.
The horizontal eye movement recordings in an individual with FIN (a) Gaze-dependent nystagmus. Note the right-beating pattern on right gaze.
- Conjugacy
- Plane of oscillations (horizontal, vertical, and torsional)
- Pattern of oscillations (pendular, jerk, or bidirectional waveforms)
- Direction of oscillations (quick phase)
- Quantitative features of the waveform, including:
- Frequency
- Amplitude
- Foveation dynamics (foveation is the period during which the eyes remain relatively still and the image is incident on the fovea)
- Null point width (range of eye eccentricities in which the nystagmus is quietest)
Note: The quantitative features of the waveform can only be evaluated using eye movement recordings.
The above measurements also help in assessing the clinical severity of the nystagmus.
- Conventionally, intensity (product of amplitude and frequency) is measured in order to describe the severity of nystagmus; however, foveation correlates best with visual function scores.
- Foveation takes into account both the retinal image velocity and position of the image in relation to the fovea. An example of the measure of foveation is the NAFX (extended nystagmus acuity function), which assesses the standard deviation of the aforementioned parameters and the duration of the foveation.
Measuring intensity, foveation characteristics, and null point width before and after treatment provides an objective measure of the therapeutic response.
Numerous studies have shown that the predominant waveform changes with age (see Table 2). In a unique case report, eye movements were described and recorded before the onset of nystagmus [Gottlob 1997].
At the onset, large-amplitude, low-frequency horizontal eye movements (described as triangular eye movements) are seen. This waveform pattern is followed by a smaller-amplitude pendular or jerk waveform and development of foveation. Another study reported that the predominant waveform during the first six months was asymmetric pendular and jerk with extended foveation [Hertle et al 2002].
Table 2.
How the Infantile Nystagmus Waveform Evolves: An Example
In adults, a pendular waveform is more commonly associated with FRMD7-related infantile nystagmus (FIN) than with non-FRMD7 idiopathic infantile nystagmus (IIN) (see Differential Diagnosis) [Thomas et al 2008]. These oscillations are accentuated by attention, anxiety, attempts to fixate on an object, and directing the gaze away from the null zone.
Individuals with FIN report good visual acuity (typically >6/12) because the nystagmus waveform is interrupted by a foveation period and, in contrast to other forms of infantile nystagmus, FIN is not the result of sensory abnormalities (e.g., reduced visual acuity resulting from foveal hypoplasia) (see Differential Diagnosis).
An abnormal head posture is seen in approximately 15% of affected individuals. Affected individuals may assume an anomalous head posture if they have an eccentric null zone. Titubation of the head is observed in some individuals. However, affected individuals do not report any tremor of the limbs or trunk or any balance or coordination issues.
Oscillopsia, the illusion of movement in one's surroundings, is very rarely reported in FIN. This may result in part from the presence of foveation periods during the waveform. However, an affected individual may complain of oscillopsia when looking at a position of gaze in which the nystagmus is more pronounced or when the individual is tired.
Affected females report slightly better visual acuity than affected males. However, no notable differences in amplitude, frequency, and waveform of nystagmus are observed between males and females.
The optokinetic response (OKR) is abnormal, with either low gains or reversal patterns described [Thomas et al 2008]. In individuals with periodic alternating nystagmus and a FRMD7 pathogenic variant no OKR is observed [Thomas et al 2008, Thomas et al 2011]. In unaffected female carriers a subnormal OKR has been described [Thomas et al 2008].
Optical coherence tomography studies have shown either normal fovea morphology or grade 1 foveal hypoplasia [Thomas et al 2014]. Similarly, optic nerve morphology can be abnormal in FIN [Thomas et al 2014, Choi et al 2018].
Genotype-Phenotype Correlations
Studies have shown extensive intra- and interfamilial variability in the phenotype [Self et al 2007, Shiels et al 2007, Thomas et al 2008].
FIN with periodic alternating nystagmus is predominantly associated with FRMD7 pathogenic missense variants [Thomas et al 2011].
Penetrance
Penetrance of FRMD7-related infantile nystagmus (FIN) is full in males and approximately 50% in females; however, eye movement recordings of clinically unaffected females can on occasion reveal a subclinical form of nystagmus [Tarpey et al 2006, Thomas et al 2008]. Analysis of eye movements to an optokinetic stimulus may show a poor response in some clinically unaffected females [Thomas et al 2008].
Nomenclature
Idiopathic infantile nystagmus (IIN) is a term used to describe nystagmus which presents at birth or infancy. There are no associated afferent or brain defects and IIN is characterized by normal electroretinograms and visual evoked potentials. If an FRMD7 pathogenic variant is identified it should be termed FRMD7-related infantile nystagmus (FIN).
Congenital motor nystagmus is an outdated term for IIN.
Infantile nystagmus syndrome (INS) is an umbrella term used to describe different forms of infantile nystagmus (with or without sensory defects) characterized by an increasing slow phase velocity.
Prevalence
The prevalence of IIN is estimated at 2:10,000 [Sarvananthan et al 2009]. Therefore, the prevalence of FIN is lower than 2:10,000.
Genetically Related (Allelic) Disorders
No phenotypes other than those discussed in this GeneReview are known to be associated with pathogenic variants in FRMD7.
Differential Diagnosis
The diagnosis of FRMD7-related infantile nystagmus (FIN) can be challenging as numerous causes of infantile nystagmus can present with conjugate horizontal oscillations of the eyes and reduced visual acuity. Using eye movement recordings one can determine whether there is a periodic component to the nystagmus, which would suggest the presence of periodic alternating nystagmus.
Idiopathic infantile nystagmus (IIN) is characterized by infantile nystagmus and reduced visual acuity in individuals without an identified molecular cause. It is not associated with any other sensory pathologies. Color vision, slit-lamp examination, electroretinogram, and visual evoked potential are normal. Strabismus is uncommon (~10% of affected individuals). Eye movement recordings show conjugate horizontal oscillations with an increasing slow phase velocity. IIN is similar to FIN; however, some of the distinguishing signs can include: abnormal head posture and eccentric null zone; amplitude of the nystagmus that is not as significantly dependent on gaze as in FIN; pendular waveform that is not as commonly encountered as in FIN.
Affected individuals rarely have a family history of nystagmus; in rare cases autosomal dominant inheritance has been reported [Klein et al 1998, Kerrison et al 1999, Hoffmann et al 2004]. See Congenital Nystagmus OMIM Phenotypic Series to view loci thought to be associated with this phenotype in OMIM.
Table 3.
Disorders to Consider in the Differential Diagnosis of FRMD7-Related Infantile Nystagmus (FIN)
Other conditions to consider. Nystagmus in childhood can also be associated with other disorders such as aniridia, retinopathy of prematurity, dystrophies of retinal photoreceptors (including Joubert syndrome and Bardet-Biedl syndrome), congenital cataract, optic disc atrophy, and optic nerve hypoplasia. Other syndromes that can present with nystagmus during infancy include Down syndrome and spasmus nutans [Gottlob 2000].
Management
Evaluations Following Initial Diagnosis
To establish the extent of disease in an individual diagnosed with FRMD7-related infantile nystagmus (FIN), the evaluations summarized in this section (if not performed as part of the evaluation that led to the diagnosis) are recommended:
- Evaluation of visual acuity at different gaze positions
- Recording eye movements to evaluate the nystagmus waveform:
- Amplitude, frequency, and conjugacy
- Foveation dynamics
- Null point width determination
- In individuals with periodic alternating nystagmus, recording of cycle duration and the presence of an alternating head posture
- Consultation with a clinical geneticist and/or genetic counselor
Treatment of Manifestations
Optical devices
- Correction of refractive errors as early as possible using contact lenses or appropriate refractive correction can improve visual acuity appreciably. Previously it was thought that contact lenses also had a role in dampening the intensity of the nystagmus. Although the mechanism is not clear, it has been suggested that dampening of the nystagmus may be exerted through the ophthalmic branch of the trigeminal nerve, which is part of the proprioceptive pathway [Dell'Osso 2002]. Recent work by Jayaramachandran et al [2014] suggest that contact lens wear does not significantly improve nystagmus or visual acuity compared to spectacle wear.
- The use of prisms may be helpful in individuals with binocular vision whose nystagmus is dampened by convergence. There are no fixed age groups for which prisms are prescribed; however, prisms are typically used in adults, teenagers, and cooperative children.
Pharmacologic. Memantine and gabapentin have been reported to improve visual acuity, intensity of nystagmus, and foveation [Shery et al 2006, McLean et al 2007].
Surgery. The Anderson-Kestenbaum procedure consists of surgery of the extraocular muscles to shift the null zone to the primary position. As mentioned in Clinical Description, the cause of an anomalous head posture is an eccentric null zone. Therefore, shifting the null zone also corrects the anomalous head posture. In practice this procedure not only shifts but also broadens the null zone, as well as decreasing nystagmus outside the null zone.
Clinical trials to assess the role of horizontal rectus tenotomy and its effects on visual function found an improvement in nystagmus waveform and visual function [Hertle et al 2003].
Surveillance
Regular follow up, especially during childhood, is necessary to evaluate for development of vision, refractive errors, strabismus, and/or amblyopia.
Evaluation of Relatives at Risk
See Genetic Counseling for issues related to testing of at-risk relatives for genetic counseling purposes.
Therapies Under Investigation
Search ClinicalTrials.gov in the US and EU Clinical Trials Register in Europe for access to information on clinical studies for a wide range of diseases and conditions. Note: There may not be clinical trials for this disorder.
Genetic Counseling
Genetic counseling is the process of providing individuals and families with information on the nature, mode(s) of inheritance, and implications of genetic disorders to help them make informed medical and personal decisions. The following section deals with genetic risk assessment and the use of family history and genetic testing to clarify genetic status for family members; it is not meant to address all personal, cultural, or ethical issues that may arise or to substitute for consultation with a genetics professional. —ED.
Mode of Inheritance
FRMD7-related infantile nystagmus (FIN) is inherited in an X-linked manner.
Risk to Family Members
Parents of a male proband
- The father of an affected male will not have the disorder nor will he be hemizygous for the FRMD7 pathogenic variant; therefore, he does not require further evaluation/testing.
- In a family with more than one affected individual, the mother of an affected male is an obligate heterozygote (carrier). Note: If a woman has more than one affected child and no other affected relatives and if the FRMD7 pathogenic variant cannot be detected in her leukocyte DNA, she most likely has germline mosaicism.
- If a male is the only affected family member (i.e., a simplex case), the mother may be a heterozygote or the affected male may have a de novo FRMD7 pathogenic variant, in which case the mother is not a carrier. Up to 7% of affected males represent simplex cases [Tarpey et al 2006].
Parents of a female proband
- A female with FRMD7-related infantile nystagmus may have inherited the pathogenic variant from either her mother or her father, or the pathogenic variant may be de novo.
- Detailed evaluation of the parents and review of the extended family history may help distinguish probands with a de novo pathogenic variant from those with an inherited pathogenic variant. Molecular genetic testing of the mother (and possibly the father, or subsequently the father) can determine if the pathogenic variant was inherited.
Sibs of a male proband. The risk to sibs depends on the genetic status of the mother:
- If the mother of the proband has an FRMD7 pathogenic variant, the chance of transmitting it in each pregnancy is 50%. Males who inherit the variant will be affected; females who inherit the variant will be heterozygotes and may or may not have nystagmus.
- If the proband represents a simplex case (i.e., a single occurrence in the family) and if the FRMD7 pathogenic variant cannot be detected in maternal leukocyte DNA, the risk to sibs is slightly greater than that of the general population (though still <1%) because of the possibility of maternal germline mosaicism.
Sibs of a female proband. The risk to the sibs depends on the genetic status of the parents:
- If the mother of the proband has an FRMD7 pathogenic variant, the chance of transmitting it in each pregnancy is 50%. Males who inherit the variant will be affected; females who inherit the variant will be heterozygotes and may or may not have nystagmus.
- If the father of the proband has an FRMD7 pathogenic variant, he will transmit the variant to all of his daughters and none of his sons.
- If the proband represents a simplex case (i.e., a single occurrence in a family) and if the FRMD7 pathogenic variant cannot be detected in the leukocyte DNA of either parent, the risk to sibs is slightly greater than that of the general population (though still <1%) because of the possibility of parental germline mosaicism.
Offspring of a proband
- Affected males will transmit the pathogenic variant to all of their daughters (who will be heterozygotes and may or may not have nystagmus) and none of their sons.
- Heterozygous females have a 50% chance of transmitting the pathogenic variant to each child.
Other family members. The risk to other family members depends on the genetic status of the proband's parents: if a parent is affected or has the pathogenic variant, the parent's family members may be at risk.
Heterozygote Detection
Molecular genetic testing of at-risk female relatives to determine their genetic status is most informative if the pathogenic variant has been identified in the proband.
Note: (1) Females who are heterozygous for this X-linked disorder may or may not have nystagmus. (2) Identification of female heterozygotes requires either (a) prior identification of the FRMD7 pathogenic variant in the family or, (b) if an affected male is not available for testing, molecular genetic testing first by sequence analysis, and if no pathogenic variant is identified, by gene-targeted deletion/duplication analysis.
Related Genetic Counseling Issues
Family planning
- The optimal time for determination of genetic risk, clarification of carrier status, and discussion of the availability of prenatal/preimplantation genetic testing is before pregnancy.
- It is appropriate to offer genetic counseling (including discussion of potential risks to offspring and reproductive options) to young adults who are affected, are carriers, or are at risk of being carriers.
Prenatal Testing and Preimplantation Genetic Testing
Once the FRMD7 pathogenic variant has been identified in an affected family member, prenatal and preimplantation genetic testing are possible.
Differences in perspective may exist among medical professionals and within families regarding the use of prenatal testing. While most centers would consider use of prenatal testing to be a personal decision, discussion of these issues may be helpful.
Resources
GeneReviews staff has selected the following disease-specific and/or umbrella support organizations and/or registries for the benefit of individuals with this disorder and their families. GeneReviews is not responsible for the information provided by other organizations. For information on selection criteria, click here.
- American Nystagmus Network, Inc.
- Nystagmus NetworkUnited KingdomPhone: 44 1427 718093Email: info@nystagmusnet.org
- Medline Plus
Molecular Genetics
Information in the Molecular Genetics and OMIM tables may differ from that elsewhere in the GeneReview: tables may contain more recent information. —ED.
Table A.
FRMD7-Related Infantile Nystagmus: Genes and Databases
Table B.
OMIM Entries for FRMD7-Related Infantile Nystagmus (View All in OMIM)
Gene structure. The longest FRMD7 transcript variant, NM_194277.2, comprises 12 exons. The length of the mRNA transcript is 3.2 kb. See Table A, Gene for a detailed summary of gene and protein information.
Pathogenic variants. More than 50 different pathogenic variants have been reported [Tarpey et al 2006, Schorderet et al 2007, Self et al 2007, Zhang et al 2007a, Zhang et al 2007b, He et al 2008, Kaplan et al 2008, Li et al 2008, Thomas et al 2011]. The pathogenic variants are distributed throughout the gene and consist of missense, nonsense, and splice site variants and frameshift deletions and insertions. Fingert et al [2010] reported the identification of an FRMD7 deletion involving exons 2, 3, and 4 in affected males from a large three-generation family with X-linked infantile nystagmus. Thomas et al [2017] reported an FRMD7 deletion of exons 2-12. AlMoallem et al [2015] reported a 1.29-Mb deletion encompassing FRMD7. The affected individual had nystagmus and autism.
Normal gene product. The gene product (FRMD7) consists of 714 amino acids with two functional domains, B41 and FERM-C. In situ hybridization experiments in human embryonic brain (~37 days post ovulation) have shown that the expression of FRMD7 is restricted to the mid- and hindbrain, regions known to be involved in motor control of eye movement [Tarpey et al 2006]. High-resolution spatial and temporal expression studies have shown that FRMD7 is expressed within the developing optokinetic and vestibuloocular reflex arcs [Thomas et al 2011, Thomas et al 2014]. Subcellular localization of FRMD7 was restricted to the neuronal cell body, the primary dendrite extension and the distal tip of the growth cone in dendrites [Betts-Henderson et al 2010]. In mice, retinal expression of FRMD7 is restricted to starburst amacrine cells [Yonehara et al 2016].
Abnormal gene product. Knockdown assays of FRMD7 in Neuro-2A cells show altered neurite outgrowth as a result of retinoic acid-induced differentiation [Betts-Henderson et al 2010]. In transiently transfected murine neuroblastoma cells no difference was seen between the subcellular localization of the mutated FRMD7 protein resulting from pathogenic missense variants and wild type. However, abnormal nuclear localization was observed with the carboxy-terminal truncated FRMD7, suggesting that the C-terminus may play a role in the subcellular localization of the FRMD7 protein [Pu et al 2011]. Four missense variants (Cys271Tyr, Gly24Glu, Arg229Cys and Ser340Leu) were studied in vitro; Cys271Tyr had the most severe cellular phenotype due to a strong dominant-negative effect on neurite outgrowth [Watkins et al 2013].
Yonehara et al [2016] demonstrated that Frmd7tm knockin mice lacked horizontal optokinetic reflex with a corresponding loss of horizontal direction selective responses in the retina.
Chapter Notes
Acknowledgments
The authors would like to acknowledge the Ulverscroft Foundation, Medical Research Council, National Institute of Health Research and Fight for Sight for their support toward research into FRMD7-related infantile nystagmus.
Author History
Irene Gottlob, MD, Univ Doz, FRCOphth (2009-present)
Michael Hisaund, BSc (2018-present)
Anil Kumar, MRCS, MRCOphth; University of Leicester (2009-2018)
Gail Maconachie, BSc, PhD (2018-present)
Frank A Proudlock, BSc, MSc, PhD; University of Leicester (2009-2018)
Mervyn G Thomas, BSc (Hons), MBChB, PhD (2009-present)
Shery Thomas, MRCOphth; University of Leicester (2009-2018)
Revision History
- 16 August 2018 (sw) Comprehensive update posted live
- 29 September 2011 (me) Comprehensive update posted live
- 12 February 2009 (me) Review posted live
- 8 October 2008 (ig) Original submission
References
Literature Cited
- AlMoallem B, Bauwens M, Walraedt S, Delbeke P, De Zaeytijd J, Kestelyn P, Meire F, Janssens S, van Cauwenbergh C, Verdin H, Hooghe S, Kumar Thakur P, Coppieters F, De Leeneer K, Devriendt K, Leroy BP, De Baere E. Novel FRMD7 mutations and genomic rearrangement expand the molecular pathogenesis of X-linked idiopathic infantile nystagmus. Invest Ophthalmol Vis Sci. 2015;56:1701–10. [PubMed: 25678693]
- Betts-Henderson J, Bartesaghi S, Crosier M, Lindsay S, Chen HL, Salomoni P, Gottlob I, Nicotera P. The nystagmus-associated FRMD7 gene regulates neuronal outgrowth and development. Hum Mol Genet. 2010;19:342–51. [PubMed: 19892780]
- Choi JH, Jung JH, Oh EH, Shin JH, Kim HS, Seo JH, Choi SY, Kim MJ, Choi HY, Lee C, Choi KD. Genotype and Phenotype Spectrum of FRMD7-Associated Infantile Nystagmus Syndrome. Invest Ophthalmol Vis Sci. 2018;59:3181–8. [PubMed: 30025138]
- Dell'Osso LF. Development of new treatments for congenital nystagmus. Ann NY Acad Sci. 2002;956:361–79. [PubMed: 11960819]
- Fingert JH, Roos B, Eyestone ME, Pham JD, Mellot ML, Stone E. Novel intragenic FRMD7 deletion in a pedigree with congenital X-linked nystagmus. Ophthalmic Genet. 2010;31:77–80. [PubMed: 20450309]
- Gottlob I. Infantile nystagmus. Development documented by eye movement recordings. Invest Ophthalmol Vis Sci. 1997;38:767–73. [PubMed: 9071231]
- Gottlob I. Nystagmus. Curr Opin Ophthalmol. 2000;11:330–5. [PubMed: 11148698]
- He X, Gu F, Wang Y, Yan J, Zhang M, Huang S, Ma X. A novel mutation in FRMD7 causing X-linked idiopathic congenital nystagmus in a large family. Mol Vis. 2008;14:56–60. [PMC free article: PMC2267738] [PubMed: 18246032]
- Hertle RW, Dell'Osso LF, FitzGibbon EJ, Thompson D, Yang D, Mellow SD. Horizontal rectus tenotomy in patients with congenital nystagmus: results in 10 adults. Ophthalmology. 2003;110:2097–105. [PubMed: 14597515]
- Hertle RW, Maldanado VK, Maybodi M, Yang D. Clinical and ocular motor analysis of the infantile nystagmus syndrome in the first 6 months of life. Br J Ophthalmol. 2002;86:670–5. [PMC free article: PMC1771154] [PubMed: 12034691]
- Hoffmann S, Becker A, Hoerle S, Metz A, Oertel WH, Sommer N, Hemmer B. Autosomal dominant congenital nystagmus is not linked to 6p12, 7p11, and 15q11 in a German family. Am J Ophthalmol. 2004;138:439–43. [PubMed: 15364228]
- Jayaramachandran P, Proudlock FA, Odedra N, Gottlob I, McLean RJ. A randomized controlled trial comparing soft contact lens and rigid gas-permeable lens wearing in infantile nystagmus. Ophthalmology. 2014;121:1827–36. [PubMed: 24811959]
- Kaplan Y, Vargel I, Kansu T, Akin B, Rohmann E, Kamaci S, Uz E, Ozcelik T, Wollnik B, Akarsu NA. Skewed X inactivation in an X linked nystagmus family resulted from a novel, p.R229G, missense mutation in the FRMD7 gene. Br J Ophthalmol. 2008;92:135–41. [PubMed: 17962394]
- Kerrison JB, Vagefi MR, Barmada MM, Maumenee IH. Congenital motor nystagmus linked to Xq26-q27. Am J Hum Genet. 1999;64:600–7. [PMC free article: PMC1377771] [PubMed: 9973299]
- Klein C, Vieregge P, Heide W, Kemper B, Hagedorn-Greiwe M, Hagenah J, Vollmer C, Breakefield XO, Kompf D, Ozelius L. Exclusion of chromosome regions 6p12 and 15q11, but not chromosome region 7p11, in a German family with autosomal dominant congenital nystagmus. Genomics. 1998;54:176–7. [PubMed: 9806847]
- Li N, Wang L, Cui L, Zhang L, Dai S, Li H, Chen X, Zhu L, Hejtmancik JF, Zhao K. Five novel mutations of the FRMD7 gene in Chinese families with X-linked infantile nystagmus. Mol Vis. 2008;14:733–8. [PMC free article: PMC2324116] [PubMed: 18431453]
- McLean R, Proudlock F, Thomas S, Degg C, Gottlob I. Congenital nystagmus: randomized, controlled, double-masked trial of memantine/gabapentin. Ann Neurol. 2007;61:130–8. [PubMed: 17279539]
- Pu J, Li Y, Liu Z, Yan Y, Tian J, Chen S, Zhang B. Expression and localization of FRMD7 in human fetal brain, and a role for F-actin. Mol Vis. 2011;17:591–7. [PMC free article: PMC3049738] [PubMed: 21386928]
- Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24. [PMC free article: PMC4544753] [PubMed: 25741868]
- Sarvananthan N, Surendran M, Roberts EO, Jain S, Thomas S, Shah N, Proudlock FA, Thompson JR, McLean RJ, Degg C, Woodruff G, Gottlob I. The prevalence of nystagmus: the Leicestershire nystagmus survey. Invest Ophthalmol Vis Sci. 2009;50:5201–6. [PubMed: 19458336]
- Schorderet DF, Tiab L, Gaillard MC, Lorenz B, Klainguti G, Kerrison JB, Traboulsi EI, Munier FL. Novel mutations in FRMD7 in X-linked congenital nystagmus Mutation in brief #963 Online. Hum Mutat. 2007;28:525. [PubMed: 17397053]
- Self JE, Shawkat F, Malpas CT, Thomas NS, Harris CM, Hodgkins PR, Chen X, Trump D, Lotery AJ. Allelic variation of the FRMD7 gene in congenital idiopathic nystagmus. Arch Ophthalmol. 2007;125:1255–63. [PubMed: 17846367]
- Shery T, Proudlock FA, Sarvananthan N, McLean RJ, Gottlob I. The effects of gabapentin and memantine in acquired and congenital nystagmus: a retrospective study. Br J Ophthalmol. 2006;90:839–43. [PMC free article: PMC1857134] [PubMed: 16556621]
- Shiels A, Bennett TM, Prince JB, Tychsen L. X-linked idiopathic infantile nystagmus associated with a missense mutation in FRMD7. Mol Vis. 2007;13:2233–41. [PubMed: 18087240]
- Tarpey P, Thomas S, Sarvananthan N, Mallya U, Lisgo S, Talbot CJ, Roberts EO, Awan M, Surendran M, McLean RJ, Reinecke RD, Langmann A, Lindner S, Koch M, Jain S, Woodruff G, Gale RP, Degg C, Droutsas K, Asproudis I, Zubcov AA, Pieh C, Veal CD, Machado RD, Backhouse OC, Baumber L, Constantinescu CS, Brodsky MC, Hunter DG, Hertle RW, Read RJ, Edkins S, O'Meara S, Parker A, Stevens C, Teague J, Wooster R, Futreal PA, Trembath RC, Stratton MR, Raymond FL, Gottlob I. Mutations in FRMD7, a newly identified member of the FERM family, cause X-linked idiopathic congenital nystagmus. Nat Genet. 2006;38:1242–4. [PMC free article: PMC2592600] [PubMed: 17013395]
- Thomas MG, Crosier M, Lindsay S, Kumar A, Thomas S, Araki M, Talbot CJ, McLean RJ, Surendran M, Taylor K, Leroy BP, Moore AT, Hunter DG, Hertle RW, Tarpey P, Langmann A, Lindner S, Brandner M, Gottlob I. The clinical and molecular genetic features of idiopathic infantile periodic alternating nystagmus. Brain. 2011;134:892–902. [PMC free article: PMC4125620] [PubMed: 21303855]
- Thomas MG, Crosier M, Lindsay S, Kumar A, Araki M, Leroy BP, McLean RJ, Sheth V, Maconachie G, Thomas S, Moore AT, Gottlob I. Abnormal retinal development associated with FRMD7 mutations. Hum Mol Genet. 2014;23:4086–93. [PMC free article: PMC4082370] [PubMed: 24688117]
- Thomas MG, Maconachie G, Sheth V, McLean RJ, Gottlob I. Development and clinical utility of a novel diagnostic nystagmus gene panel using targeted next-generation sequencing. Eur J Hum Genet. 2017;25:725–34. [PMC free article: PMC5477371] [PubMed: 28378818]
- Thomas S, Proudlock FA, Sarvananthan N, Roberts EO, Awan M, McLean R, Surendran M, Kumar AS, Farooq SJ, Degg C, Gale RP, Reinecke RD, Woodruff G, Langmann A, Lindner S, Jain S, Tarpey P, Lucy Raymond F, Gottlob I. Phenotypical characteristics of idiopathic infantile nystagmus with and without mutations in FRMD7. Brain. 2008;131:1259–67. [PubMed: 18372314]
- Watkins RJ, Patil R, Goult BT, Thomas MG, Gottlob I, Shackleton S. A novel interaction between FRMD7 and CASK: evidence for a causal role in idiopathic infantile nystagmus. Hum Mol Genet. 2013;22:2105–18. [PMC free article: PMC3633374] [PubMed: 23406872]
- Yonehara K, Fiscella M, Drinnenberg A, Esposti F, Trenholm S, Krol J, Franke F, Scherf BG, Kusnyerik A, Müller J, Szabo A, Jüttner J, Cordoba F, Reddy AP, Németh J, Nagy ZZ, Munier F, Hierlemann A, Roska B. Congenital nystagmus gene FRMD7 is necessary for establishing a neuronal circuit asymmetry for direction selectivity. Neuron. 2016;89:177–93. [PMC free article: PMC4712192] [PubMed: 26711119]
- Zhang B, Liu Z, Zhao G, Xie X, Yin X, Hu Z, Xu S, Li Q, Song F, Tian J, Luo W, Ding M, Yin J, Xia K, Xia J. Novel mutations of the FRMD7 gene in X-linked congenital motor nystagmus. Mol Vis. 2007a;13:1674–9. [PubMed: 17893669]
- Zhang Q, Xiao X, Li S, Guo X. FRMD7 mutations in Chinese families with X-linked congenital motor nystagmus. Mol Vis. 2007b;13:1375–8. [PubMed: 17768376]
Publication Details
Author Information and Affiliations
University of Leicester
Leicester, United Kingdom
University of Leicester
Leicester, United Kingdom
University of Leicester
Leicester, United Kingdom
University of Leicester
Leicester, United Kingdom
Publication History
Initial Posting: February 12, 2009; Last Update: August 16, 2018.
Copyright
GeneReviews® chapters are owned by the University of Washington. Permission is hereby granted to reproduce, distribute, and translate copies of content materials for noncommercial research purposes only, provided that (i) credit for source (http://www.genereviews.org/) and copyright (© 1993-2024 University of Washington) are included with each copy; (ii) a link to the original material is provided whenever the material is published elsewhere on the Web; and (iii) reproducers, distributors, and/or translators comply with the GeneReviews® Copyright Notice and Usage Disclaimer. No further modifications are allowed. For clarity, excerpts of GeneReviews chapters for use in lab reports and clinic notes are a permitted use.
For more information, see the GeneReviews® Copyright Notice and Usage Disclaimer.
For questions regarding permissions or whether a specified use is allowed, contact: ude.wu@tssamda.
Publisher
University of Washington, Seattle, Seattle (WA)
NLM Citation
Thomas MG, Maconachie G, Hisaund M, et al. FRMD7-Related Infantile Nystagmus. 2009 Feb 12 [Updated 2018 Aug 16]. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2024.